Research
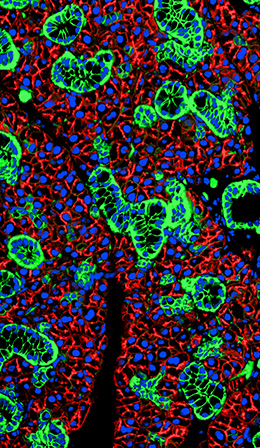
Overview
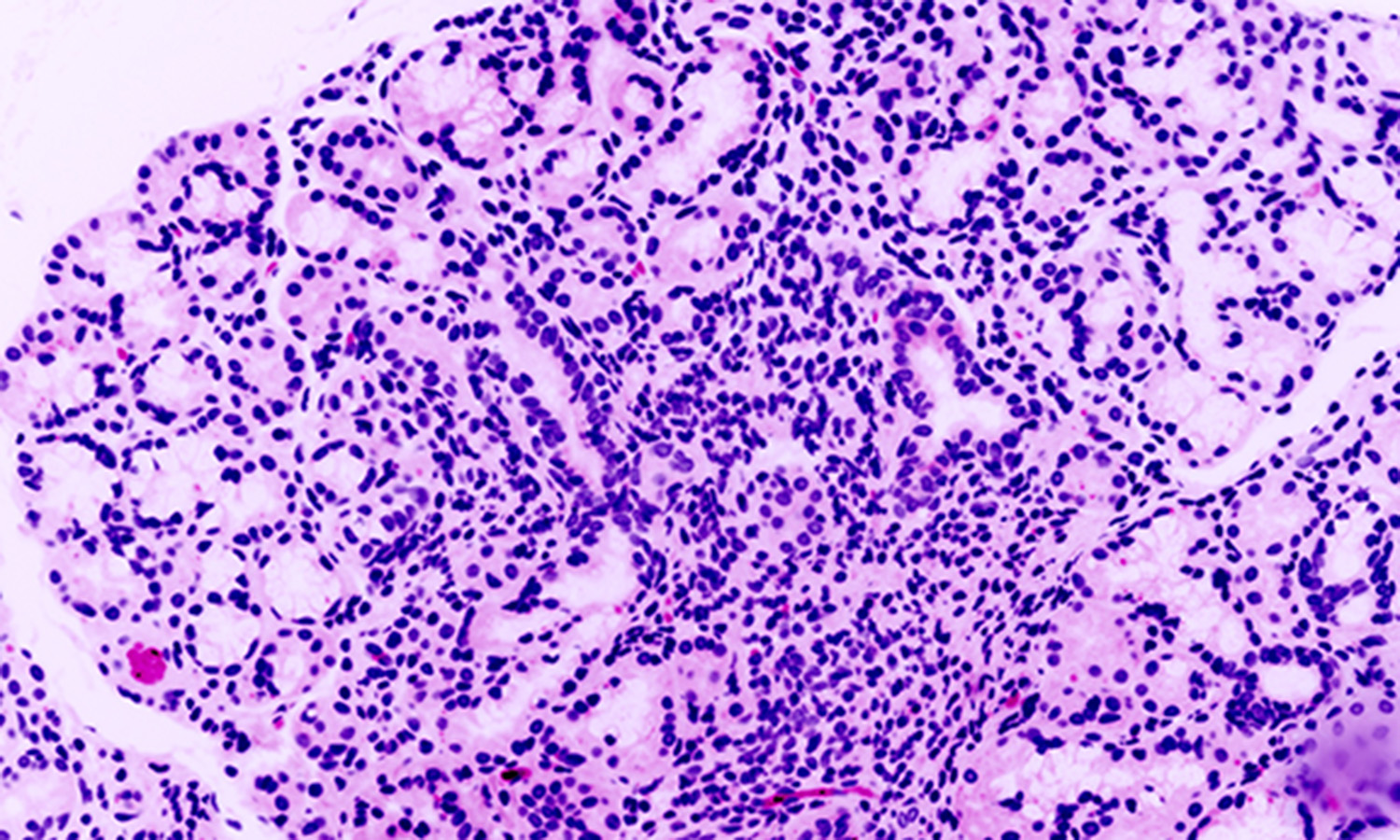
My laboratory is focused on understanding how salivary glands develop in the embryo and understanding disease progression for informing development of future therapeutics. Fibrillar proteins, which are components of the extracellular matrix, which is found in the extracellular space surrounding cells are critical for development of the structure of the salivary gland and for influencing their function. In response to injury, there is an increase in the production of extracellular matrix. Although extracellular matrix remodeling is a natural response to injury, excessive ECM deposition, or fibrosis, limits regeneration, is a causative factor in hundreds of diseases, and leads to up to 45% of all deaths worldwide. Fibrosis occurs in salivary glands of patients treated with radiation for head and neck cancers and in patients suffering from the autoimmune disease, Sjögren’s Syndrome. Despite the known inhibitory effects of fibrosis on tissue regeneration, and involvement of fibrosis in disease, the mechanisms through which fibrosis develops in the salivary gland and leads to dysfunction have not been explored. Our lab is using injury models for human disease and single cell RNA sequencing methods to define the cells that are critical for injury and that become misregulated with fibrotic disease. We are using organoids, which are miniature organs that grow in a dish, to examine the contribution of cells that are found in the connective tissue compartment of organs and that contribute to fibrosis and recovery from fibrosis. In addition, we are exploring whether manipulation of signaling in cells that contribute to fibrosis can reverse or delay development of fibrosis and can restore organ function in injury models.
Other projects
Organoid Differentitation
Organoids are useful tools for understanding cell-cell signaling and modeling disease. We are using knowledge of development to engineer organoids that better mimic organs in vivo to better understand developmentl processes and to develop organoids as tools to study disease development.
Endothelial Cells
Endothelial cells of the vasulature are criticla for salivary gland development and are aberrant in Sjogren’s Syndrome disease. Endothelial cells make factors known as angiocrine factors that promote regenerative processes. We are identifying angiocrine factors that may have therapeutic applications.
Tissue Engineering
Scaffolds can facilitate the transfer of therapeutic cells for regenerative therapies. With collaborators who generate scaffolds we are testing the efficacy of scaffolds that mimic native extracellular matrix for delivery of mesenchymal stem cell-like cells for remediation of fibrotic disease.
Contact
Melinda Larsen
mlarsen@albany.edu
518-591-8882